 In this clock the induction period is the time it takes for autocatalytic process to start after which the concentration of free iodine falls rapidly as observed by UV/VIS spectroscopy.In an iodine clock reaction, colour changes after a time delay.Ī chemical clock (or clock reaction) is a complex mixture of reacting chemical compounds in which the onset of an observable property (discoloration or coloration) occurs after a predictable induction time due to the presence of clock species at a detectable amount. I 3 - → I - + I 2 chlorate ion oxidizes iodide ion to hypoiodous acid and Chlorous acid in slow and rate-determining step:ĬlO 3 - + I - + 2H + → HOI + HClO 2 chlorate consumption is accelerated by reaction of hypoiodous acid to iodous acid and more chlorous acid.ĬlO 3 - + HOI + H + → HIO 2 + HClO 2 more autocatalysis when newly generated iodous acid also converts chlorate in the fastest reaction step. iodide anions present in equilibrium with triiodide ion and iodine: Ammonium persulfate has a higher solubility and is used instead in reaction document examples from Oxford University.Īn experimental iodine clock sequence has also been established for a system consisting of iodine potassium-iodide, sodium chlorate and perchloric acid that takes place through the following reactions. Potassium persulfate has a low solute potential, according to documents on the Salters website. Once all the thiosulphate is consumed the Iodine may form a complex with the starch. Sodium thiosulfate is used to reduce Iodine back to Iodide before the Iodine can complex with the starch to form the characteristic blue-black colour.ĢI - (aq) + S 2O 8 2- (aq) → I 2 (aq) + 2SO 4 2- (aq) And is then removed: This clock reaction uses sodium, potassium or ammonium persulfate to oxidise iodide ions to iodine. When the bisulphite is fully consumed, the iodine will survive (i.e., no reduction by the bisulfite) to form the dark blue complex with starch. However, the iodine is reduced immediately back to iodide by the bisulfite: The iodate in excess will oxidize the iodide generated above to form iodine: In this protocol, iodide ion is generated by the following slow reaction between the iodate and bisulfite: Adding more thiosulfate will have the opposite effect it will take longer for the blue color to appear.Īn alternative protocol uses a solution of iodate ion (for instance potassium iodate) to which an acidified solution (again with sulfuric acid) of sodium bisulfite is added. Increasing the pH, or the concentration of iodide or hydrogen peroxide will shorten the time. Once the thiosulfate ion has been exhausted, this reaction stops and the blue color caused by the triiodide - starch complex appears.Īnything that accelerates the first reaction will shorten the time until the solution changes color. When the solutions are mixed, the second reaction causes the triiodide ion to be consumed much faster than it is generated, and only a small amount of triiodide is present in the dynamic equilibrium. 
I 3 -(aq) + 2 S 2O 3 2-(aq) → 3 I -(aq) + S 4O 6 2-(aq)Īfter some time the solution will rapidly change color to a very dark blue, almost black. In the second, fast reaction, triiodide is reconverted to iodide by the thiosulfate. 
In the first, slow reaction, the triiodide ion is produced. 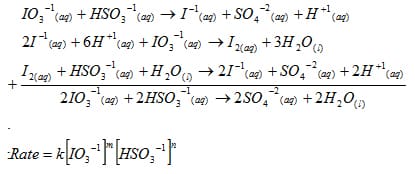
There are two reactions occurring in the solution. To this is added a solution containing potassium iodide, sodium thiosulfate, and starch. This reaction starts from a solution of hydrogen peroxide with sulfuric acid.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
